 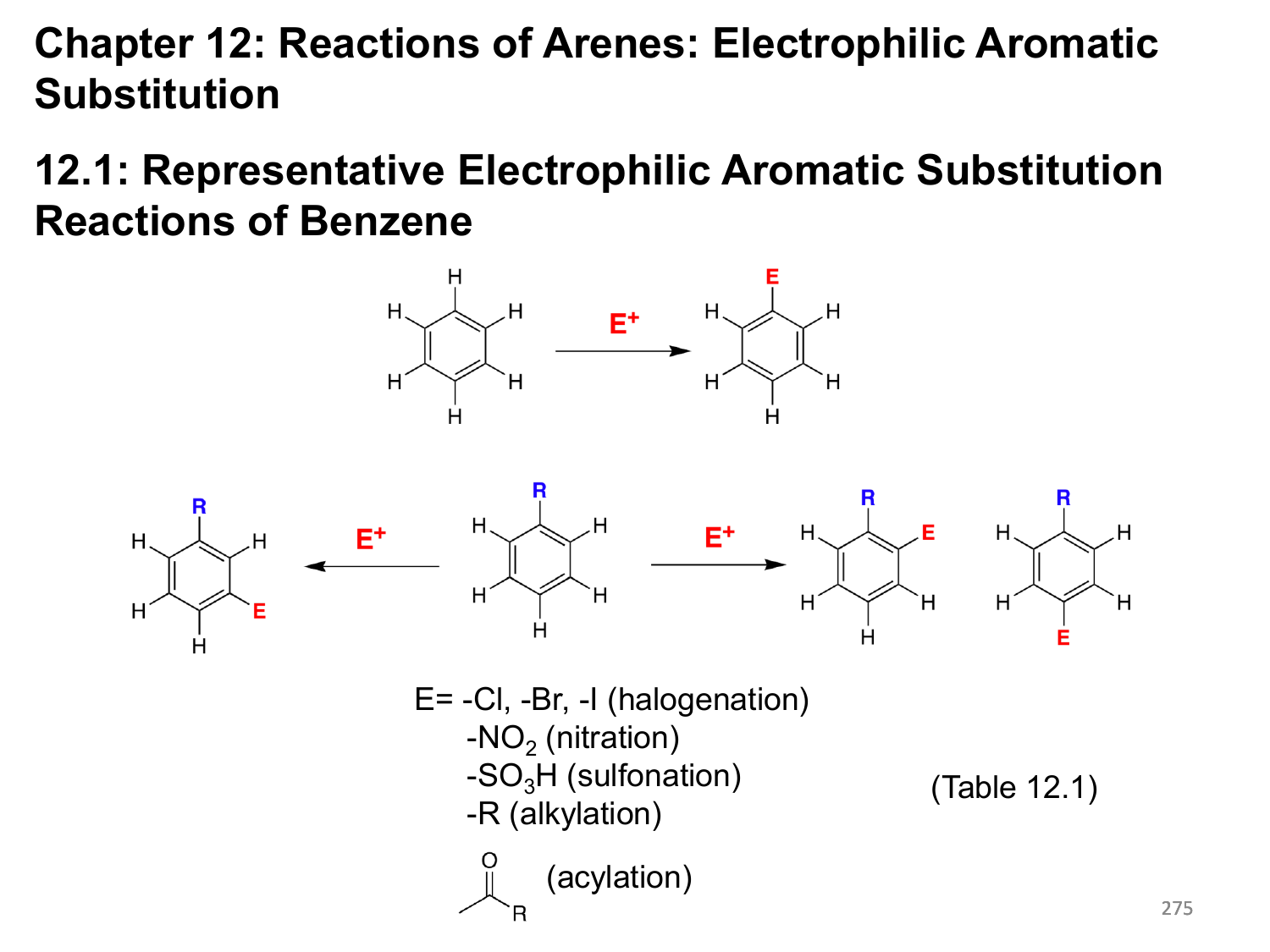
Energy Diagram of Electrophilic Aromatic Substitution Reaction Remember that the positively charged carbon atom loses a proton here as an electrophile is charging the benzene ring. This reaction is a quick method and typically exergonic. Some of the critical factors that affect the electrophile reaction here are steric hindrance and chance. Due to the loss of aromaticity, there is a high energy activator there, and this reaction is endergonic. This process is a very time taking and slow method. The electrophile attacks the aromatic ring. This particle primarily has three resonance contributors. It leads to the development of charged and delocalised cyclohexadienyl ions. The reaction starts with the electrophile attacking the pi-electron present in the aromatic benzene ring. The mechanism mainly has two steps mentioned below. So this is the substitute of a hydrogen atom with an electrophile linked with a benzene ring. So electrophilic aromatic substitution is a reaction in which an electrophile replaces an atom linked with an aromatic ring. A purposeful cluster may be a substituent that brings bound chemical reactions that the hydrocarbon itself does not show. Numerous other functional groups can be added to the compound by this reaction. 
Electrophilic Aromatic Substitution Reaction These catalysts are either Bronsted-Lowry acids or Lewis acids. Such a large amount of EAS reactions require a catalyst to activate the electrophile. Many electrophiles (such as Br 2 ) don’t seem to be sufficiently electrophilic to react independently. The second step regenerates the aromatic system, which is a quicker step. However, it is still a lot less stable than the beginning material this loss of aromaticity implies that the primary step is often the speed determinant step in EAS. The Wheland intermediate is stable by resonance. It is a carbocation intermediate consisting of five sp 2 -hybridised C atoms and one sp 3 -hybridised C atom.This intermediate then loses H + through electrophilic elimination. The product of Step 1 (i.e., addition of the electrophile) is called an arenium ion intermediate or a Wheland intermediate. The mechanism of electrophilic aromatic substitution of benzene involves two steps. In other words we can say that there is a substitution of an electrophile in place of a hydrogen of an aromatic compound. In electrophilic aromatic substitution of benzene, an electrophile reacts with an aromatic compound. Benzene undergoes electrophilic substitution reactions that preserve aromaticity, rather than electrophilic addition reactions (the reactions characteristic of alkenes), which would destroy aromaticity. In the substitution reaction, an electrophile substitutes for one of the hydrogens attached to the benzene ring. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
